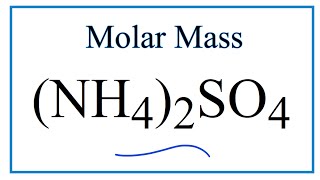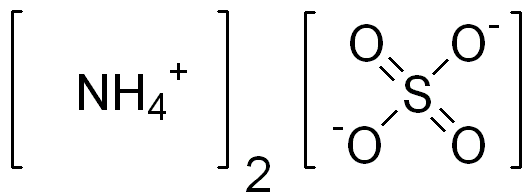SOLVED: A solution is prepared by dissolving 25.0 g of ammonium sulfate in enough water to make 100.0 mL of stock solution. A 10.00 mL sample of this stock solution is added
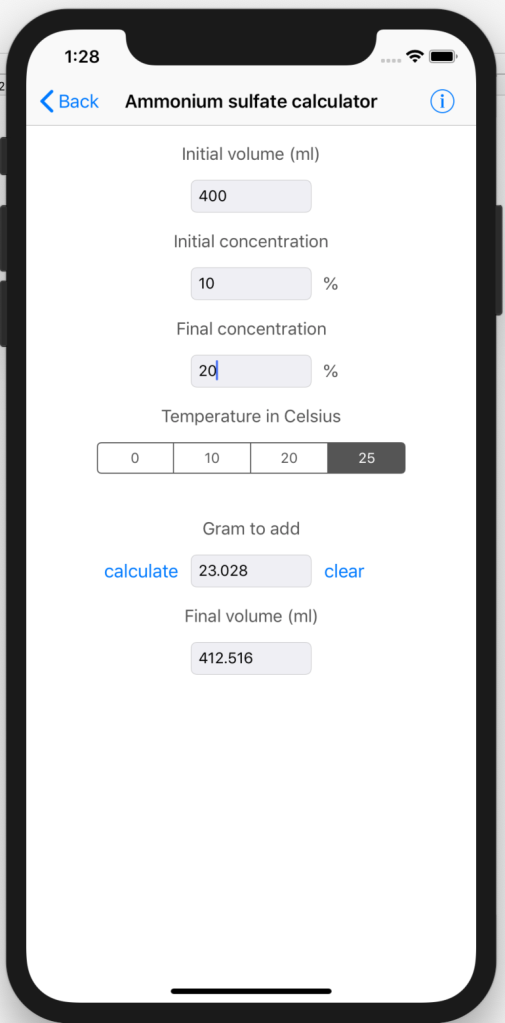
GitHub - fomightez/small_scale_ammonium_sulfate_precipitation_calculator: A tool facilitating designing a pilot experiment to screen for protein precipitation conditions.
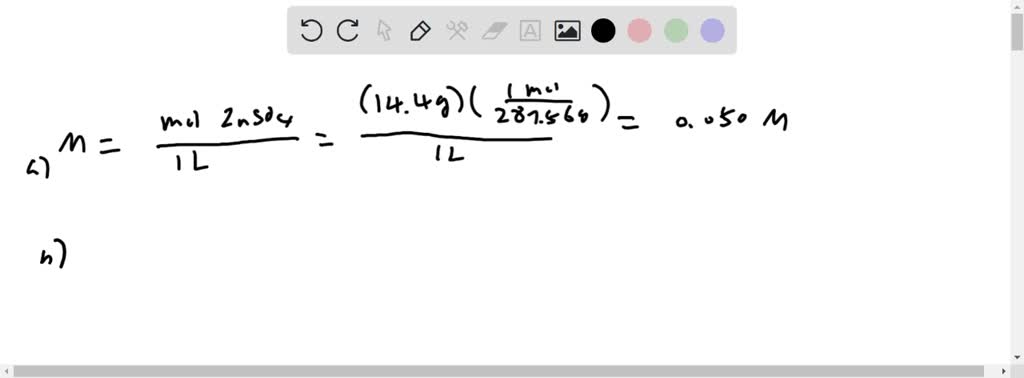
SOLVED: Calculate the molarity of 14.4 g ZnSO4 (MW 287.56 g/mol) dissolved to make 1 L of the solution: How many volumes (in mL) of 0.05 M zinc sulfate is expected to
![Ammonium Phosphate [(NH4)3PO4] - Structure, Molecular Mass, Properties and Uses of Ammonium Phosphate Ammonium Phosphate [(NH4)3PO4] - Structure, Molecular Mass, Properties and Uses of Ammonium Phosphate](https://cdn1.byjus.com/wp-content/uploads/2021/03/Ammonium-Phosphate-NH43PO4-700x300.png)
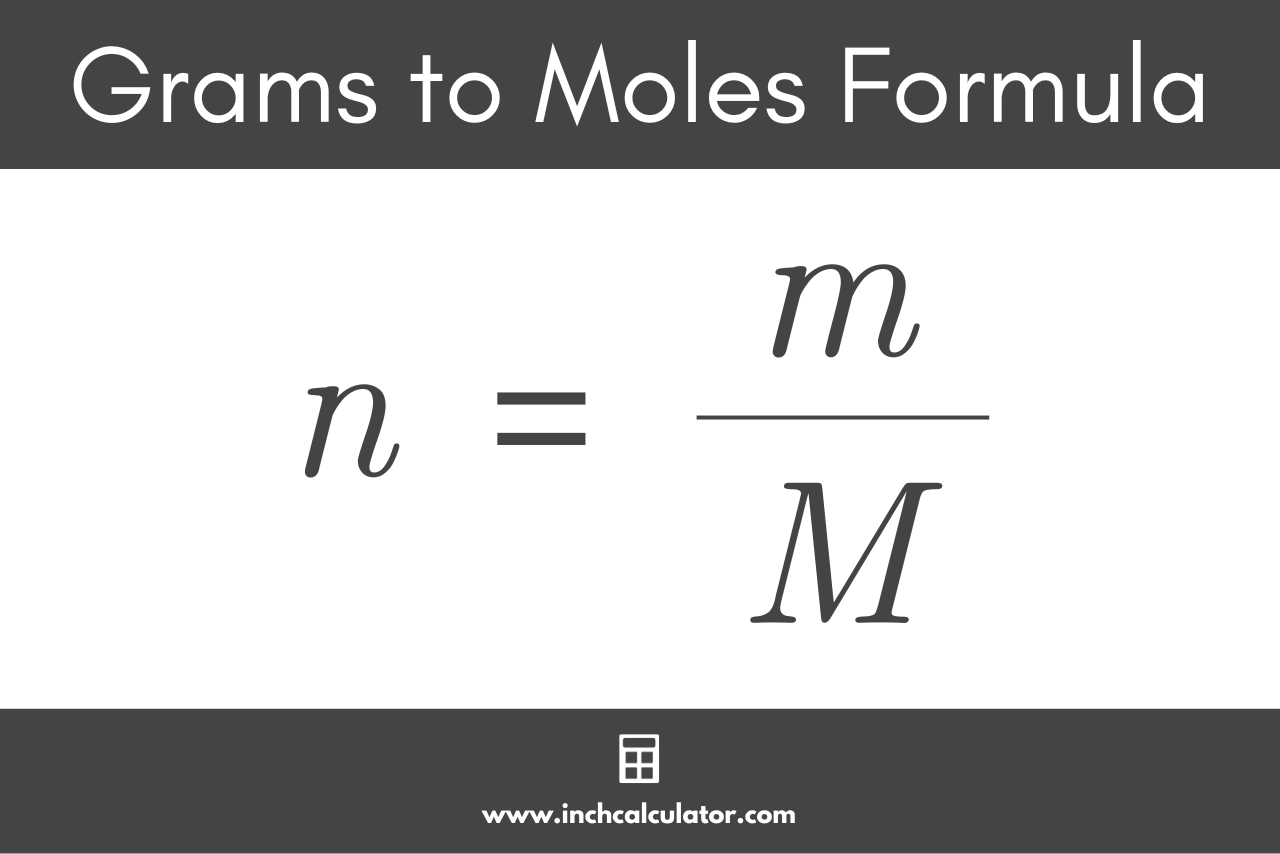

![Ammonium Oxide [(NH4)2O] Molecular Weight Calculation - Laboratory Notes Ammonium Oxide [(NH4)2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonium-oxide-molecular-weight-calculation-300x176.jpg)

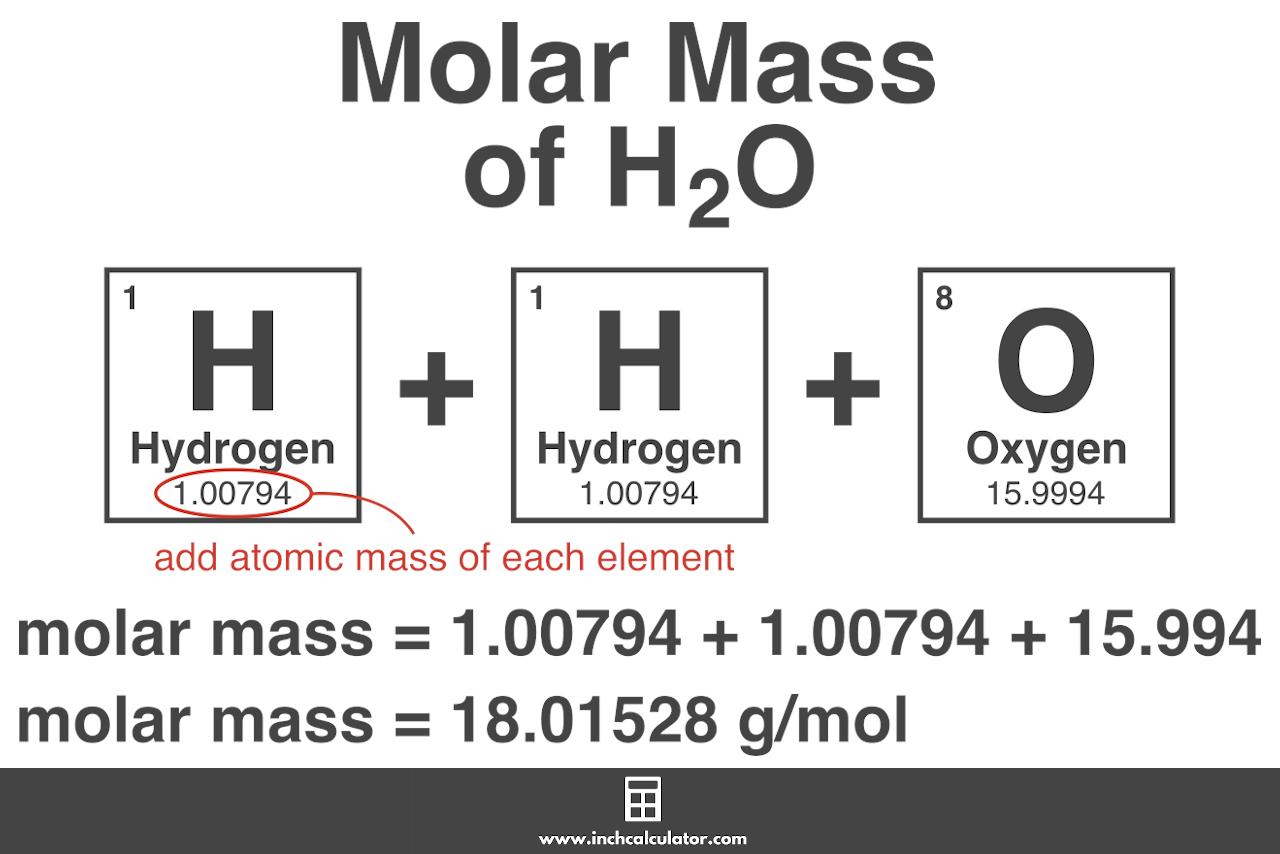
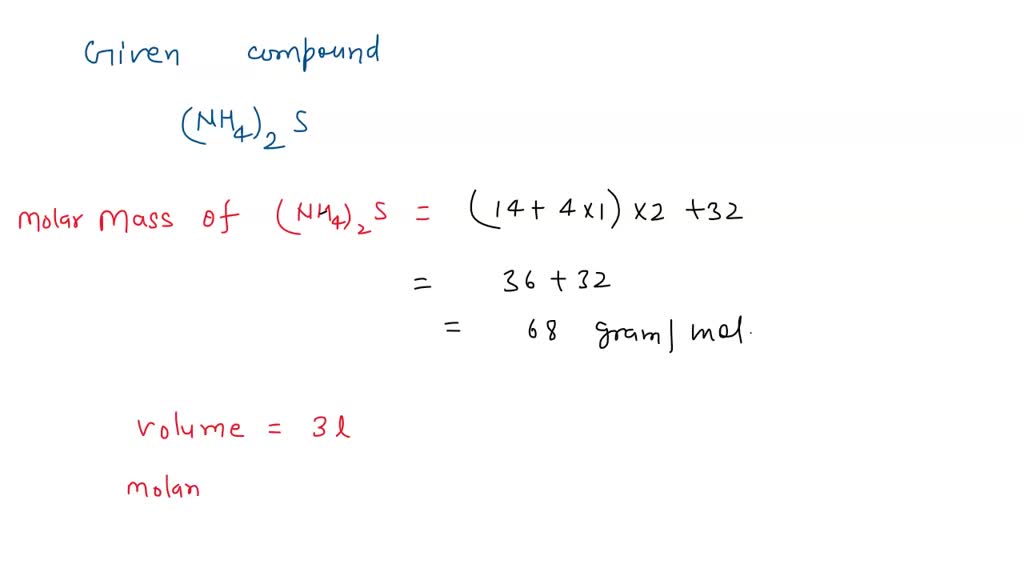


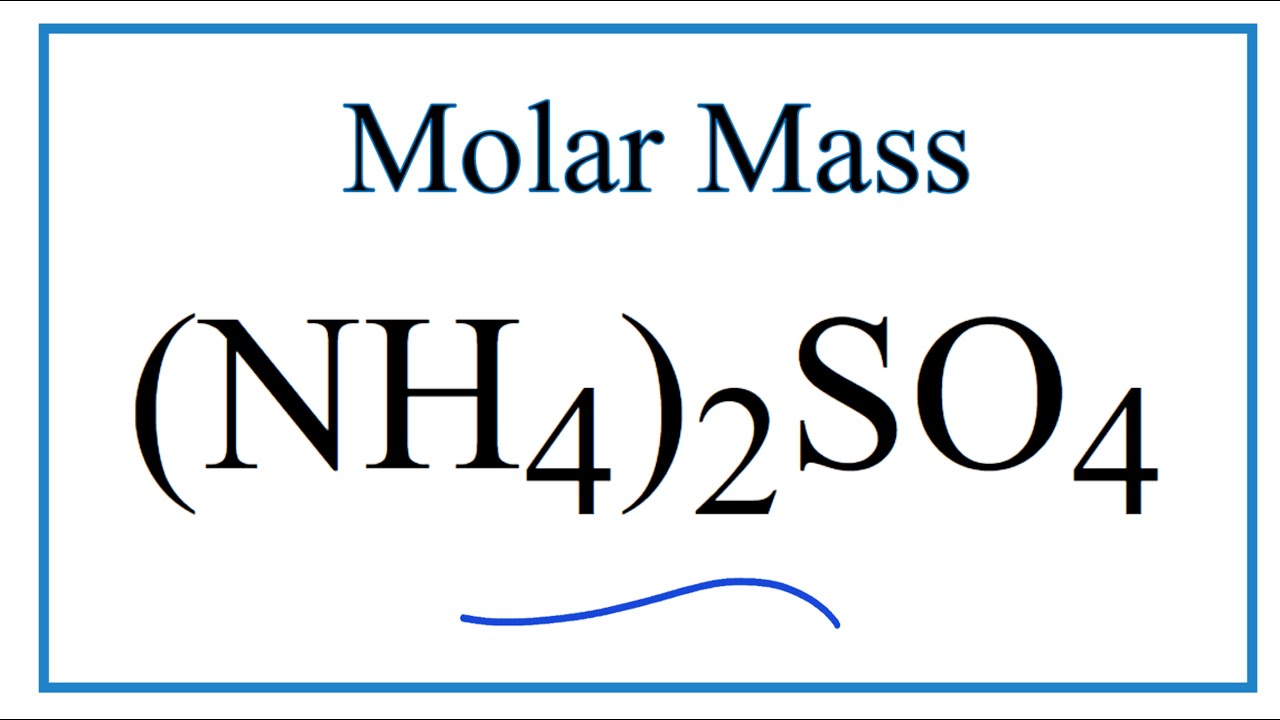
![Ammonium Sulfate [(NH4)2SO4] Molecular Weight Calculation - Laboratory Notes Ammonium Sulfate [(NH4)2SO4] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/ammonium-sulfate-molecular-weight-calculation-300x204.jpg)