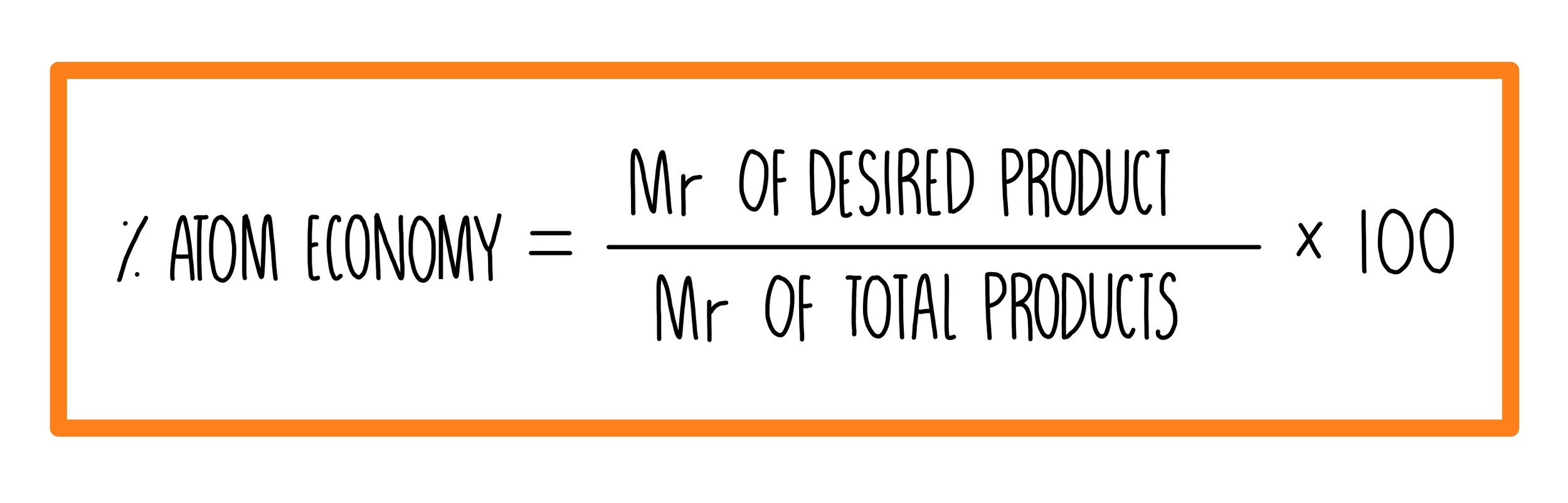
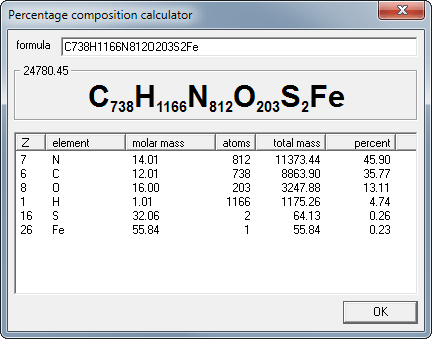
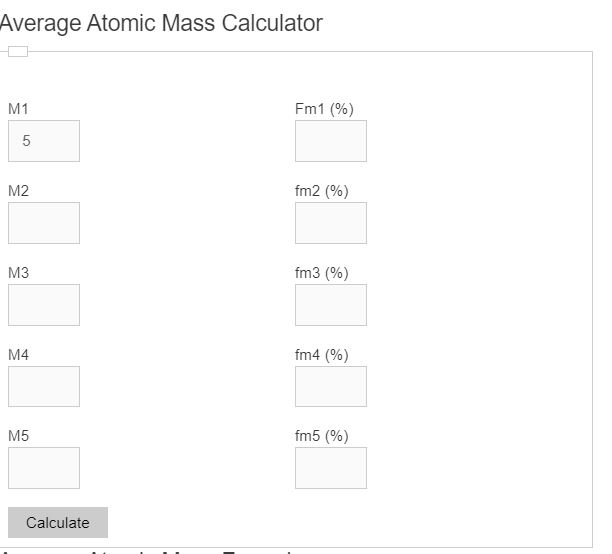
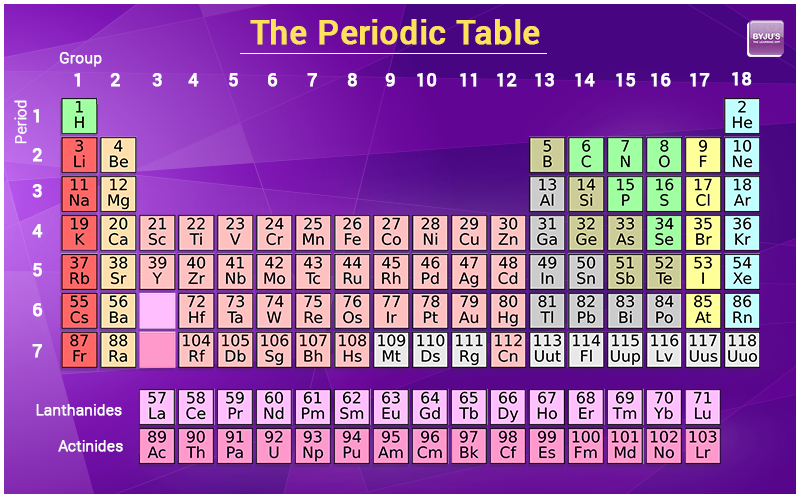
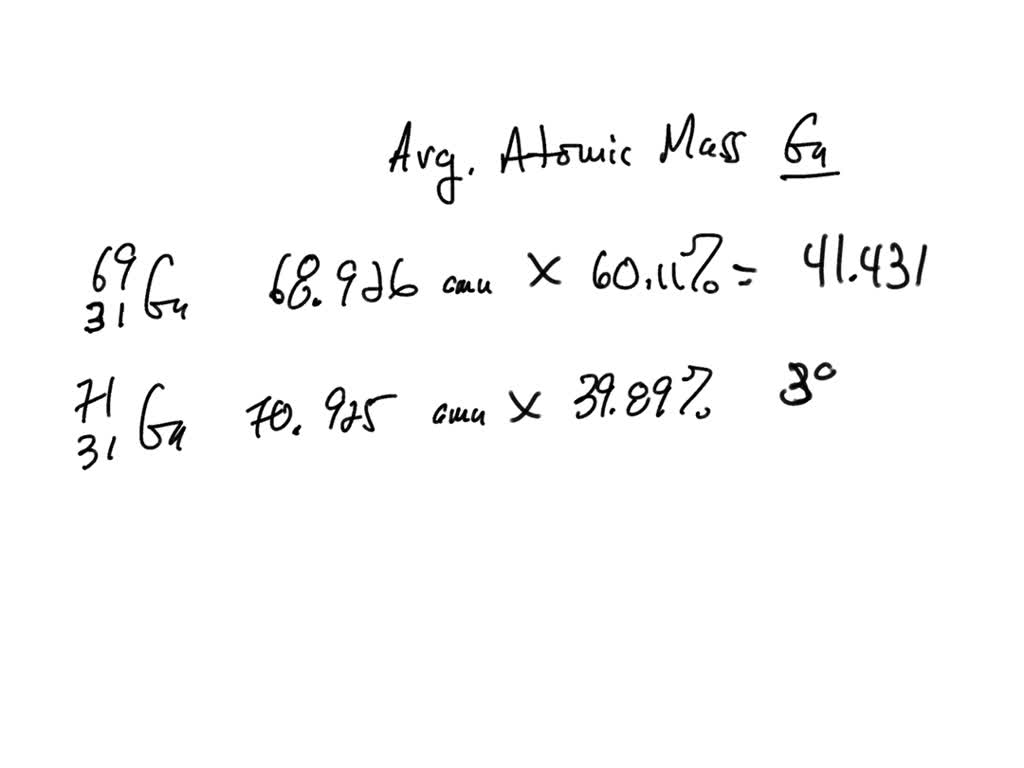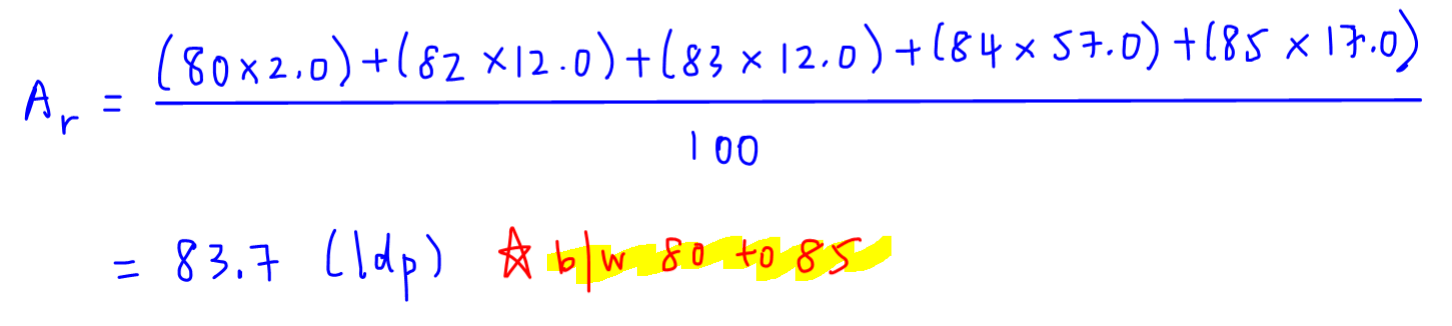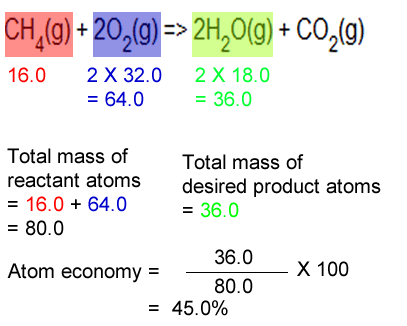materials - How do you derive the formula for converting weight percent to atom percent? - Engineering Stack Exchange

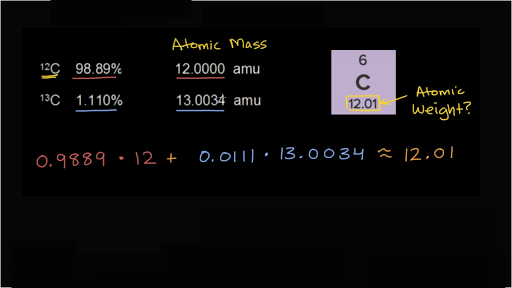
a) The percentage abundance of isotope 10 Ne is 90% and that of isotope 22Ne is 100% Calculate the average atomic mass of Neon. trong in the atom of the olomon