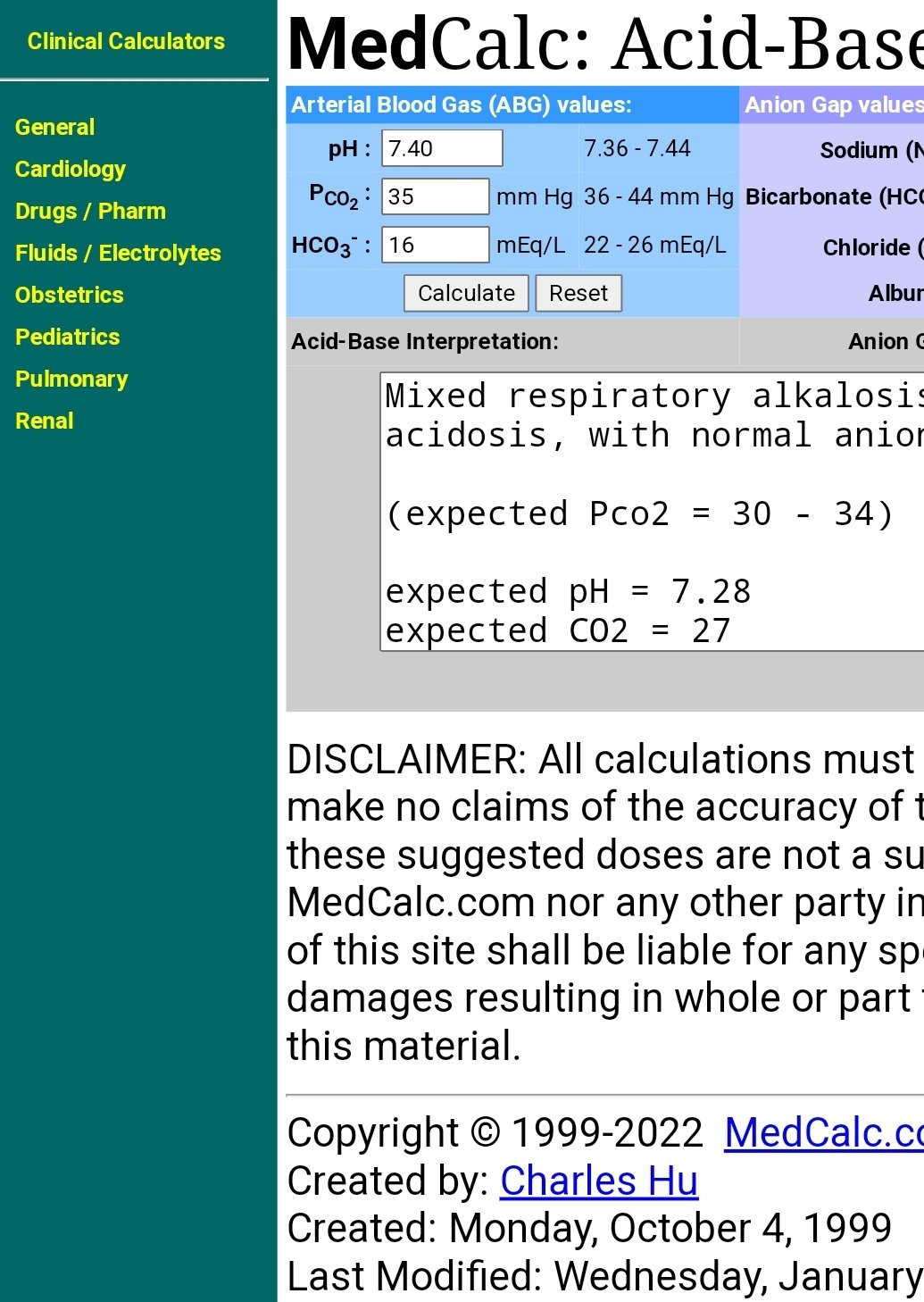
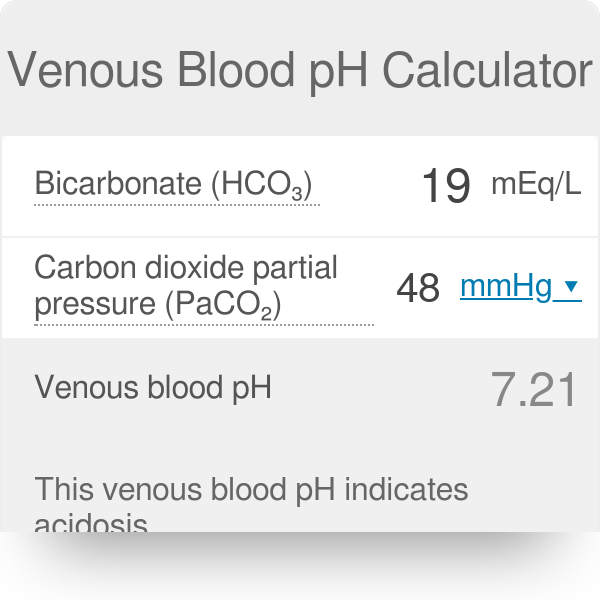
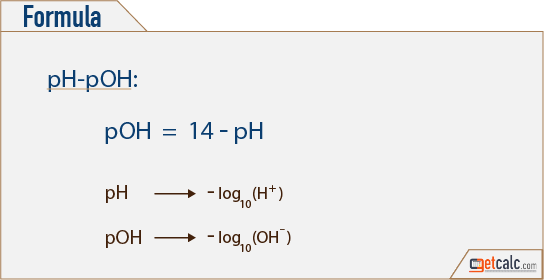
SOLVED: Calculate the pH of a blood plasma sample with a total CO2 concentration of 26.9 mM and bicarbonate concentration of 25.6 mM. Recall from page 67 that the relevant pK2 of
![The `pH` of blood is 7.4 . What is the ratio of `[(HPO_(4)^(2-))/(H_(2)PO_(4)^(-))]` in the blood. ` - YouTube The `pH` of blood is 7.4 . What is the ratio of `[(HPO_(4)^(2-))/(H_(2)PO_(4)^(-))]` in the blood. ` - YouTube](https://i.ytimg.com/vi/irWUJj_qIt8/maxresdefault.jpg)
The `pH` of blood is 7.4 . What is the ratio of `[(HPO_(4)^(2-))/(H_(2)PO_(4)^(-))]` in the blood. ` - YouTube