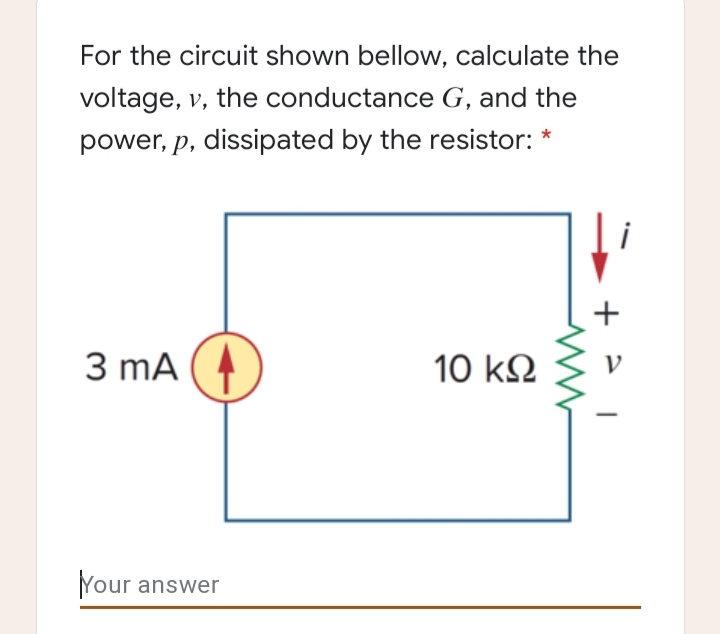
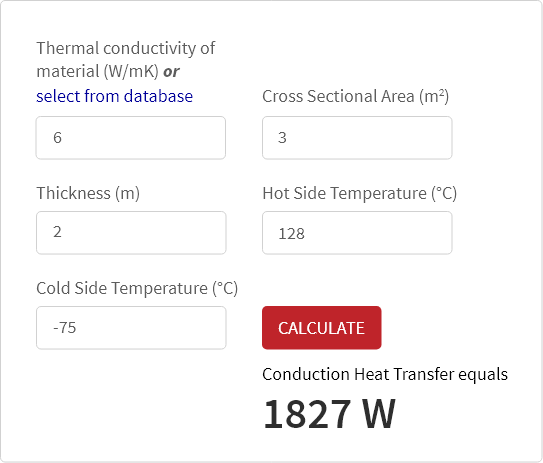
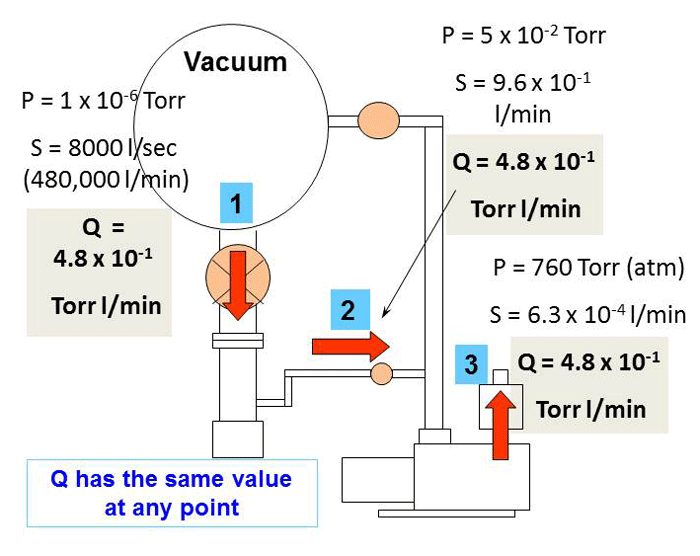
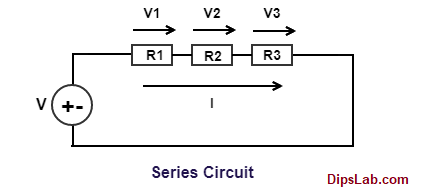
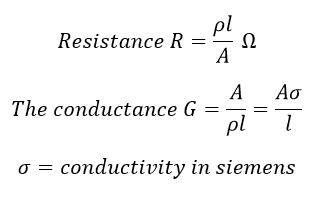The specific conductance of a solution containing 5 g of anhydrous BaCl2 (mol. wt. = 208) in 1000 cm^3 of a solution is found to be 0.0058 ohm^-1 cm^-1 . Calculate the

Specific conductance of pure water at `25^(@)C` is `0.58 xx 10^(-7)` mho `cm^(-1)`. Calculate - YouTube
Setup for the calculation of the differential conductance in a bilayer... | Download Scientific Diagram

calculate the molar conductance of 001m solution of an electrolyte which has a resistance of 210ohms at 298k cell constant is 088cm 1 g2znqh44 -Chemistry - TopperLearning.com
![Conductance and Resistance] What does the little o and p mean, and when im rearranging do i need to move them away from A and L or not? : r/PhysicsStudents Conductance and Resistance] What does the little o and p mean, and when im rearranging do i need to move them away from A and L or not? : r/PhysicsStudents](https://i.redd.it/8vzkx4bt4dk51.png)
Conductance and Resistance] What does the little o and p mean, and when im rearranging do i need to move them away from A and L or not? : r/PhysicsStudents

Calculate the equivalent conductance at infinite dilution of the salt NaKC2O4 . Given ionic equivalent conductance of oxalate, Na^ + and k^ + ions are 74.1, 50.1 and 73.5 S cm^2 eq^-1
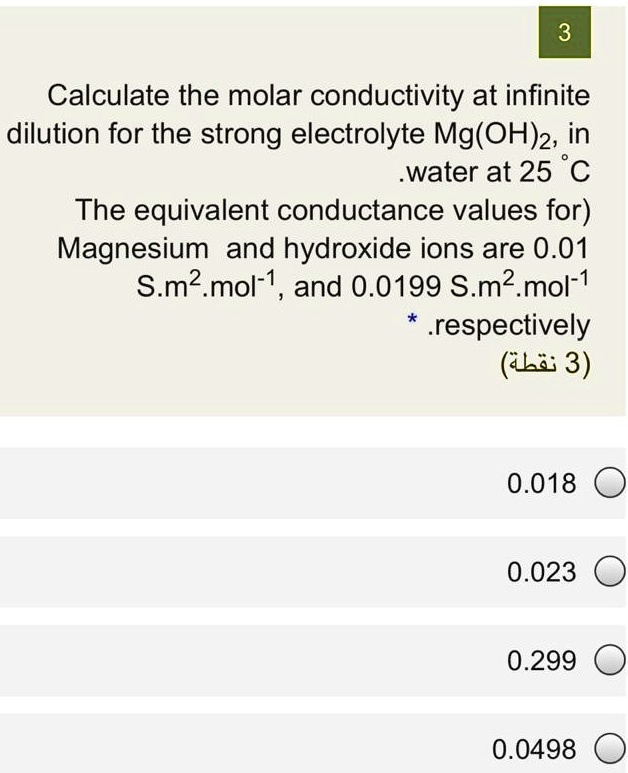
SOLVED: 3 Calculate the molar conductivity at infinite dilution for the strong electrolyte Mg(OH)2; in water at 25 C The equivalent conductance values for) Magnesium and hydroxide ions are 0.01 S.m2. mol-1 ,