
Percent Yield and Percent Error Calculations | Science, Chemistry, Percent Yield, percent error | ShowMe

SOLVED: Calculate percent recovery for each dye value: extract using the grape Kool-Aid spectrum as the starting point. Generally, percent recovery is calculated using this formula: % recovery = (recovered amount /

SOLVED: If you had started with 0.929 g of solid copper and ended with 0.882 g, what is the percent recovery? (Show calculation) What could happen in the experiment to cause this? (

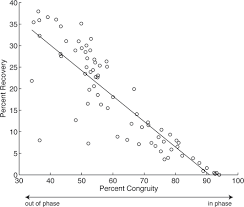




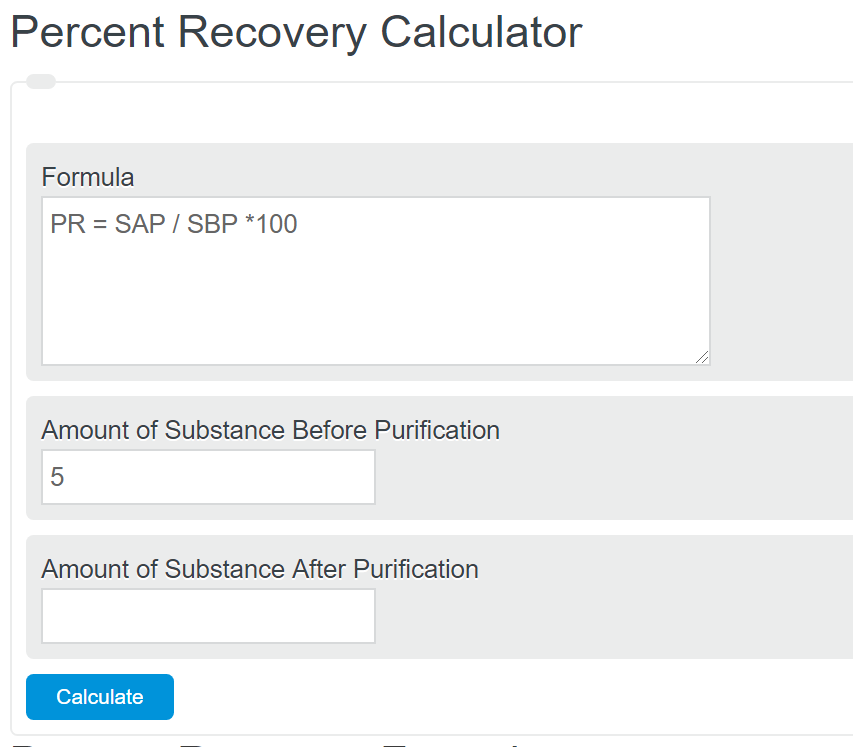










:max_bytes(150000):strip_icc()/recovery-rate.asp_Final-a8fac2c32d704c628b7408edd9604684.png)